When a patient shows signs of dehydration, the problem isn’t just thirst—it’s happening at the cellular level. A patient is in need of hydration. Which type of solution are the patient’s cells most likely in? The answer: a hypertonic environment.
In simple terms, the fluid outside the cells has a higher concentration of solutes than the fluid inside. This causes water to move out of the cells, leaving them shrunken and stressed. Understanding this concept is essential for students, healthcare workers, and anyone interested in how the body maintains fluid balance.
Let’s break it down in plain language.
Understanding Tonicity and Cell Hydration
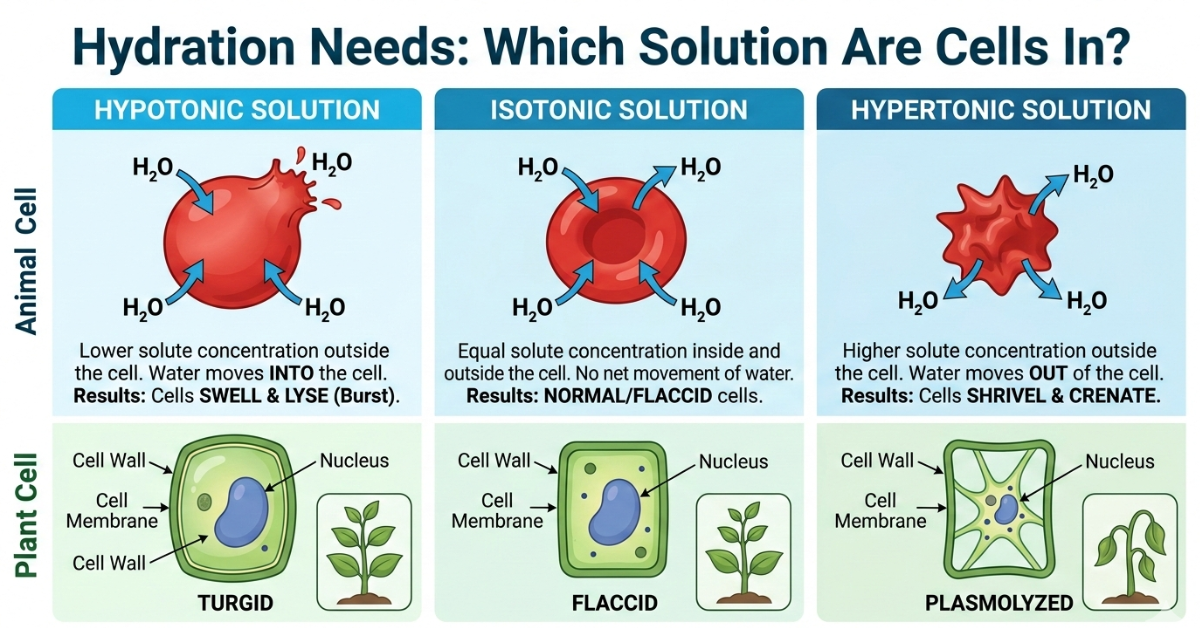
Tonicity describes the concentration of solutes (like sodium and electrolytes) outside the cell compared to inside. Water always moves toward the area with a higher solute concentration—a process called osmosis.
There are three main types of solutions:
1. Isotonic Solution
-
Equal solute concentration inside and outside the cell
-
No net movement of water
-
Cells maintain normal size and function
2. Hypotonic Solution
-
Lower solute concentration outside the cell
-
Water moves into the cell
-
Cells swell and may burst
3. Hypertonic Solution (Key Concept)
-
Higher solute concentration outside the cell
-
Water moves out of the cell
-
Cells shrink (crenate)
When a patient needs hydration, their cells are most likely in a hypertonic solution.
Why Dehydration Creates a Hypertonic Environment
Dehydration occurs when the body loses more water than it takes in. This loss concentrates electrolytes—especially sodium—in the extracellular fluid.
Common causes include:
-
Excessive sweating
-
Vomiting or diarrhea
-
Fever
-
Poor fluid intake
-
Severe burns
-
Certain medications (like diuretics)
As extracellular fluid becomes more concentrated:
-
Osmotic pressure increases
-
Water leaves the cells
-
Cells shrink and function poorly
This cellular dehydration is what drives symptoms like:
-
Dry mouth
-
Fatigue
-
Confusion
-
Low blood pressure
-
Rapid heart rate
What Happens to Cells in a Hypertonic Solution?
When cells are surrounded by a hypertonic fluid:
-
Water exits the cell through osmosis
-
Cell volume decreases
-
Metabolic processes slow down
-
In severe cases, cells may be damaged
Red blood cells, for example, become shriveled—a process known as crenation. In the brain, cellular dehydration can lead to neurological symptoms such as dizziness or confusion.
This is why prompt and appropriate fluid replacement is critical.
How Healthcare Providers Correct the Problem
Treatment focuses on restoring fluid balance and returning the extracellular environment to isotonic.
Common Hydration Strategies
Oral Rehydration (mild cases)
-
Water
-
Oral rehydration solutions (ORS)
-
Electrolyte drinks
Intravenous Fluids (moderate to severe cases)
-
Isotonic saline (0.9% NaCl) – first-line treatment
-
Lactated Ringer’s solution
-
Hypotonic fluids (used carefully in specific situations)
The goal is simple: dilute the extracellular fluid so water can move back into the cells.
Quick Comparison: Cell Response to Solutions
| Solution Type | Water Movement | Cell Effect |
| Hypotonic | Into the cell | Swelling |
| Isotonic | No movement | Normal |
| Hypertonic | Out of the cell | Shrinking |
If a patient requires hydration, their cells are most likely shrinking due to a hypertonic environment.
Real-Life Example
Think of a marathon runner who collapses after a race without adequate fluid intake. Sweat removes water faster than electrolytes, concentrating sodium in the blood. The extracellular fluid becomes hypertonic, pulling water out of cells—especially dangerous for brain cells.
Hydration restores balance and protects cellular function.
FAQs
Q1: When a patient is dehydrated, are their cells in a hypotonic or hypertonic solution?
Hypertonic. The extracellular fluid is more concentrated, causing water to leave the cells.
Q2: Why not give hypotonic fluids immediately?
Rapid shifts into cells—especially brain cells—can cause swelling and complications. Treatment must be controlled.
Q3: What is the safest initial IV fluid for dehydration?
Isotonic saline (0.9% NaCl) is commonly used to stabilize fluid balance.
Q4: Can dehydration affect the brain?
Yes. Cellular dehydration in the brain can lead to confusion, dizziness, or even seizures in severe cases.
Q5: How do I know if dehydration is severe?
Signs include:
-
Very little urine
-
Rapid pulse
-
Low blood pressure
-
Altered mental status
Seek medical care immediately.
Conclusion
If you’re asking, “A patient is in need of hydration. Which type of solution are the patient’s cells most likely in?” the correct answer is a hypertonic solution. Dehydration concentrates the extracellular fluid, pulling water out of cells and impairing their function.
The medical response focuses on restoring balance—usually with isotonic fluids—so water can return where it belongs. Understanding this simple principle of tonicity isn’t just exam material; it’s the foundation of safe and effective patient care.